Dear ETPN members, dear colleagues, we are glad to share with you what we consider as a milestone in the preparation of a smarter Regulatory Framework for Nanomedicine brought by the REFINE Project, now available in open access as a special issue published this September 2022 in the Drug Delivery & Translation Research journal. Guest Editors: Kathleen Spring, Klaus-M. Weltring, Adriele Prina-Mello, and Ruth Schmid. This special issue brought by REFINE is a great set of available knowledge, tools and good practices to make your nanomedicine innovation ready for the clinic by design. We warmly encourage you to read it carefully and share in your own networks.
Context
The number of new designs for medicinal products and medical devices that are based on nano(bio)materials (NBMs) is increasing rapidly and along such diverse lines that current regulatory testing will efectively become a bottleneck for innovation. The current regulation, initially designed for small chemical entities, is valid but its practical application to NBMs raises several analytical and experimental difculties already in the preclinical assessment, which leads to a lack of confdence in the testing data. Regulators are aware of this danger and have started to get into a more intense dialogue with the scientifc community and with developers of NBMs, to adapt the existing regulatory strategies. It is clear, that there will not be a disruptive replacement of the existing regulatory framework, but that there must be refnements to improve safety, cost/time efciency, and sustainability. To support this refnement the partners of the REFINE consortium joint forces to pioneer a regulatory science framework (RSF) for the risk–beneft assessment of NBM-based medicinal products and medical devices.
Contents
Subcellular detection of PEBCA particles in macrophages: combining darkfield microscopy, confocal Raman microscopy, and ToF–SIMS analysis
Authors (first, second and last of 6)
A multistep in vitro hemocompatibility testing protocol recapitulating the foreign body reaction to nanocarriers
Authors (first, second and last of 6)
A Decision Support System for preclinical assessment of nanomaterials in medical products: the REFINE DSS
Authors (first, second and last of 11)
A comparative biodistribution study of polymeric and lipid-based nanoparticles
Authors (first, second and last of 12)
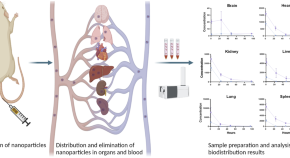
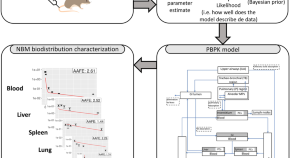
Physiologically based pharmacokinetic modeling of intravenously administered nanoformulated substances
Authors (first, second and last of 5)
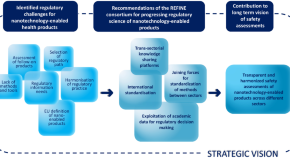
Future perspectives for advancing regulatory science of nanotechnology-enabled health products
Authors (first, second and last of 10)
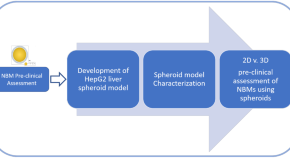
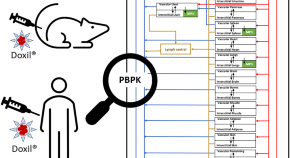
A physiologically based pharmacokinetic model to predict pegylated liposomal doxorubicin disposition in rats and human
Authors (first, second and last of 9)
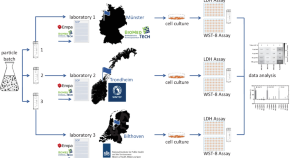
Standardization of an in vitro assay matrix to assess cytotoxicity of organic nanocarriers: a pilot interlaboratory comparison
Authors (first, second and last of 10)
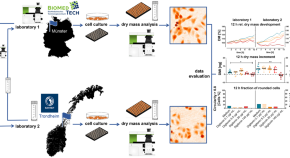
Interlaboratory evaluation of a digital holographic microscopy–based assay for label-free in vitro cytotoxicity testing of polymeric nanocarriers
Authors (first, second and last of 11)
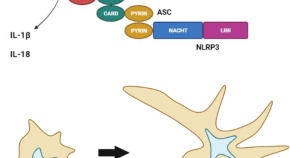
An inter-laboratory comparison of an NLRP3 inflammasome activation assay and dendritic cell maturation assay using a nanostructured lipid carrier and a polymeric nanomedicine, as exemplars
Authors (first, second and last of 4)
Evaluating nanobiomaterial-induced DNA strand breaks using the alkaline comet assay
Authors (first, second and last of 5)
Correction: Evaluating nanobiomaterial-induced DNA strand breaks using the alkaline comet assay
Authors (first, second and last of 5)
About the REFINE Project
REFINE proposes a Regulatory Science Framework for the risk-benefit assessment of medical products and medical devices that are based on nanomedicines and biomaterials. REFINE approach is aligned with the industrial R&D innovation and developmental practice. REFINE has received funding from the European Union’s Horizon 2020 Research and Innovation Programme under Grant Agreement No 761104 Learn more about the project on its website: http://refine-nanomed.eu/

